Neurotization of the facial nerve as an effective way to treat paralysis of mimic muscles
- Authors: Skaliitchouk B.V.1, Gaivoronsky A.I.1,2, Vinogradov V.V.1, Svistov D.V.1
-
Affiliations:
- Military Medical Academy named after S.M. Kirov
- St. Petersburg State University
- Issue: Vol 104, No 1 (2023)
- Pages: 134-143
- Section: Clinical experiences
- Submitted: 24.12.2021
- Accepted: 29.09.2022
- Published: 01.02.2023
- URL: https://kazanmedjournal.ru/kazanmedj/article/view/90970
- DOI: https://doi.org/10.17816/KMJ90970
- ID: 90970
Cite item
Abstract
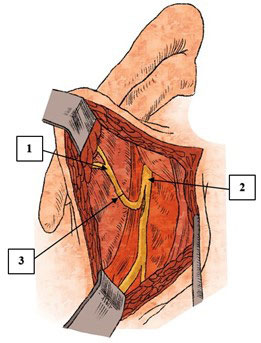
Pathologies of the facial nerve are one of the most common types of pathology of the peripheral nervous system. In the structure of lesions of the cranial nerves, this pathology occupies the first place. The clinical picture of facial nerve damage of various genesis is rather monotonous and manifests itself as persistent paresis or paralysis of the facial muscles. The literature describes a large number of different highly effective techniques aimed at restoring the function of the facial nerve and mimic muscles, examples of which are numerous conservative and surgical methods for the treatment of facial nerve neuropathy. The review presents the most common method of mimic muscles paralysis surgical treatment — facial nerve neurotization. The essence of this surgical intervention is in suturing to the affected facial nerve the trunk or a portion of individual fibers of the intact nerve-neurotic, which can be the hypoglossal, masticatory, phrenic, accessory, glossopharyngeal nerves, as well as the descending branch of the hypoglossal nerve and the anterior branches of the C2–C3 cervical nerves. Currently, options for the combined use of various donor nerves and autoextensions are gaining popularity among neurosurgeons, due to more favorable results in restoring the function of the facial nerve, as well as with an individual approach to each patient. The main stages of neurotization of the facial nerve include the isolation and intersection of the facial nerve, the isolation and intersection of the trunk or individual fibers of the neurotizer, the execution of the suture of the nerve in the “end to end” or “end to side” method. Particular attention should be paid to the most innovative method of facial nerve neurotization — facial nerve cross-plasty, during which an anastomosis between the damaged and intact facial nerves using autotransplants from the gastrocnemius nerve or a free muscle graft, including the tender muscle and the anterior branch of the obturator nerve is performed. The process of restoring facial nerve function and regressing characteristic symptoms takes a long period of time and requires specialized restorative treatment.
Full Text
About the authors
Bogdan V. Skaliitchouk
Military Medical Academy named after S.M. Kirov
Author for correspondence.
Email: bogdan_skaliitchouk@mail.ru
ORCID iD: 0000-0002-6024-8142
student
Russian Federation, St. Petersburg, RussiaAlexey I. Gaivoronsky
Military Medical Academy named after S.M. Kirov; St. Petersburg State University
Email: don-gaivoronsky@ya.ru
ORCID iD: 0000-0003-1886-5486
M.D., D. Sci. (Med.), Prof., Depart. of Neurosurgery
Russian Federation, St. Petersburg, Russia; St. Petersburg, RussiaVyacheslav V. Vinogradov
Military Medical Academy named after S.M. Kirov
Email: ulytreack@gmail.com
ORCID iD: 0000-0001-5930-3805
student
Russian Federation, St. Petersburg, RussiaDmitriy V. Svistov
Military Medical Academy named after S.M. Kirov
Email: dvsvistov@mail.ru
ORCID iD: 0000-0002-3922-9887
M.D., Cand. Sci. (Med.), Assoc. Prof., Head of Depart., Depart. of Neurosurgery
Russian Federation, St. Petersburg, RussiaReferences
- Sektsiya StAR “Assotsiaciya chelyustno-litsevykh khirurgov i khirurgov-stomatologov”. Klinicheskiy protokol meditsinskoy pomoshchi patsientam s neyropatiey litsevogo nerva. (Section STAR “Association of Maxillofacial Surgeons and Dental Surgeons”. Clinical protocol of medical care for patients with neuropathy of the facial nerve.) M.; 2014. 39 р. (In Russ.)
- Reich SG. Bell's Palsy. Continuum (Minneap Minn). 2017;23(2, Selected Topics in Outpatient Neurology):447–466. doi: 10.1212/CON.0000000000000447.
- Danny J, Revenaugh PC. Facial reanimation: an update on nerve transfers in facial paralysis. Curr Opin Otolaryngol Head Neck Surg. 2019;27:231–236. doi: 10.1097/MOO.0000000000000543.
- Spencer CR, Irving RM. Causes and management of facial nerve palsy. Br J Hosp Med (Lond). 2016;77(12):686–691. doi: 10.12968/hmed.2016.77.12.686.
- Chang YS, Choi JE, Kim SW, Baek SY, Cho YS. Prevalence and associated factors of facial palsy and lifestyle characteristics: Data from the Korean National Health and Nutrition Examination Survey 2010–2012. BMJ Open. 2016;6(11):e012628. doi: 10.1136/bmjopen-2016-012628.
- Tsymbaliuk IaV, Tsymbaliuk VI, Tretyak IB, Medvediev VV, Gurianov VG, Gatskiy AA, Petriv TI. Comparative analysis of various types of neurotization as a method of surgical treatment of peripheral facial paresis. Novosti Khirurgii. 2020;28(3):299–308. (In Russ.) doi: 10.18484/2305-0047.2020.3.299.
- Hohman MH, Hadlock TA. Etiology, diagnosis, and management of facial palsy: 2000 patients at a facial nerve center. Laryngoscope. 2014;124:E283–E293. doi: 10.1002/lary.24542.
- Gordin E, Lee TS, Ducic Y, Arnaoutakis D. Facial nerve trauma: evaluation and considerations in management. Craniomaxillofac Trauma Reconstr. 2015;8(1):1–13. doi: 10.1055/s-0034-137252.
- Shurgaya CM, Nerobeev AI, Karayan AC, Myaskovskaya EM. Possibilities of neuroneurotization in the treatment of patients with facial paralysis. Stomatologiya. 1995;74(4):33–39. (In Russ.)
- Kalakutskiy NV, Pakhomova NV, Petropavlovskaya OYu. Algoritm of the choice of various options of surgical techniques of treatment of patients with tumors parotid salivary gland and paralysis of mimic muscles. Mezhdunarodnyy zhurnal prikladnykh i fundamentalnykh issledovaniy. 2016;(12-9):1637–1640. (In Russ.)
- Grebenyuk VI, Chuprina YuV. Khirurgicheskoe lechenie paralichey litsevykh myshts. (Surgical treatment of paralysis of the facial muscles.) L.: Meditsina; 1964. 155 р. (In Russ.)
- Zotov AV, Rzayev DA, Dmitriev AB, Chernov SV, Moysak GI. Evaluation of short-term surgical outcomes in facial paralysis patients treated by trigeminal neurotization. Voprosy neyrohirurgii. 2016;(4):31–39. (In Russ.) doi: 10.17116/neiro201680431-39.
- Zlotnik EI, Sklyut IA, Smeyanovich AF, Korotkeviya EA. Litsevoy nerv v khirurgii nevrinom sluhovogo nerva. (The facial nerve in surgery for acoustic neuromas.) Minsk: Belarus; 1978. 1543 р. (In Russ.)
- Kaverina VV. Regeneratsiya nervov pri neyroplasticheskikh operatsiyakh. (Nerve regeneration in neuroplastic surgery.) L.: Meditsina; 1975. 215 р. (In Russ.)
- Brown S, Isaacson B, Kutz W, Barnett S, Rozen SM. Facial nerve trauma: Clinical evaluation and management strategies. Plast Reconstruct Surg. 2019;143(5):1498–1512. doi: 10.1097/PRS.0000000000005572.
- Goven'ko FS. Khirurgiya povrezhdeniya perifericheskikh nervov. (Surgery for damage to peripheral nerves.) Saint-Petersburg: Feniks; 2010. 384 p. (In Russ.)
- Tsymbaliuk IV, Tretyak IB, Gatskiy OO, Luzan BM, Petriv TI, Tsymbaliuk VI. Differentiated methods for surgical treatment of patients with facial nerve injury consequences. Travma. 2019;20(6):45–52 (In Ukr.) doi: 10.22141/1608-1706.6.20.2019.186034.
- Matos Cruz AJ. Facial nerve repair. Treasure Island (FL): StatPearls Publishing; 2021. https://www.ncbi.nlm.nih.gov/books/NBK560623/ (access date: 14.11.2021).
- Sánchez-Ocando M, Gavilán J, Penarrocha J, González-Otero T, Moraleda S, Roda JM, Lassaletta L. Facial nerve repair: the impact of technical variations on the final outcome. Eur Arch Otorhinolaryngol. 2019; 276:3301–3308. doi: 10.1007/s00405-019-05638-8.
- Tarek A. Amer, Mohamed S. El Kholy. The split hypoglossal nerve versus the cross-face nerve graft to supply the free functional muscle transfer for facial reanimation: A comparative study. J Plast Reconstr Aesthet Surg. 2018;71:750–757. doi: 10.1016/j.bjps.2018.01.008.
- Nechaeva AS, Ulitin AYu, Pustovoy SV, Tastanbekov MM. Hypoglossal-facial nerve anastomosis for management of facial palsy. Russian neurosurgical journal named after professor AL Polenov. 2019;21(3):32–37. (In Russ.)
- Socolovsky M, Roberto S, Masi G. Treatment of complete facial palsy in adults: comparative study between direct hemihypoglossal-facial neurorrhaphy, hemihipoglossal-facial neurorrhaphy with grafts, and masseter to facial nerve transfer. Acta Neurochir. 2016;158;945–957. doi: 10.1007/s00701–016–2767–7.
- Yetiser S. Immediate hypoglossal-facial anastomosis in patients with facial interruption. J Craniofac Surg. 2018;29(3):648–650. doi: 10.1097/SCS.0000000000004150.
- Samii M, Alimohamadi M, Khouzani RK, Rashid MR, Gerganov V. Comparison of direct side-to-end and end-to-end hypoglossal-facial anastomosis for facial nerve repair. World Neurosurg. 2015;84(2):368–375. doi: 10.1016/j.wneu.2015.03.029.
- Venail F, Sabatier P, Mondain M, Segniarbieux F, Leipp C, Uziel A. Outcomes and complications of direct end-to-side facial-hypoglossal nerve anastomosis according to the modified May technique. J Neurosurg. 2009;110(4):786–791. doi: 10.3171/2008.9.JNS08769.
- Dziedzic TA, Kunert P, Marchel A. Hemihypoglossal-facial nerve anastomosis for facial nerve reanimation: Case series and technical note. World Neurosurg. 2018;118:460–467. doi: 10.1016/j.wneu.2018.06.217.
- Gao Z. Neurorrhaphy for facial reanimation with interpositional graft: Outcome in 23 patients and the impact of timing on the outcome. World Neurosurgy. 2019;19:1–16 doi: 10.1016/j.wneu.2019.02.124.
- Biglioli F, Frigerio A, Colombo V, Colletti G, Rabbiosi D, Mortini P, Dalla Toffola E, Lozza A, Brusati R. Masseteric-facial nerve anastomosis for early facial reanimation. Craniomaxillofac Surg. 2012;40(2):149–155. doi: 10.1016/j.jcms.2011.03.005.
- Hontanilla B, Marre D, Cabello A. Masseteric nerve for reanimation of the smile in short-term facial paralysis. Br J Oral Maxillofac Surgery. 2014;52(2);118–123. doi: 10.1016/j.bjoms.2013.09.017.
- Collar RM, Byrne PJ, Boahene KD. The subzygomatic triangle: Rapid, minimally invasive identification of the masseteric nerve for facial reanimation. Plast Reconstr Surg. 2013;132(1):183–188. doi: 10.1097/prs.0b013e318290f6dc.
- Boahene K. Reanimating the paralyzed face. F1000 Prime Rep. 2013;5–49. doi: 10.12703/P5-49.
- Fournier HD, Denis F, Papon X, Hentati N, Mercier P. An anatomical study of the motor distribution of the mandibular nerve for a masseteric-facial anastomosis to restore facial function. Surg Radiol Anat. 1997;19:241–244. doi: 10.1007/BF01627866.
- Gayvoronsky IV, Rodionov AA, Gayvoronskaya MG, Nichiporuk GI, Shashkov VA. Role of chewing muscles and temporomandibular joint in the realization of mandibula buttress function. Bulletin of the Russian Military medical academy. 2017;60(4):158–163. (In Russ.)
- Albathi M, Oyer S, Ishii LE, Byrne P, Ishii M, Boahene KO. Early nerve grafting for facial paralysis after cerebellopontine angle tumor resection with preserved facial nerve continuity. JAMA Facial Plast Surg. 2016;18(1);54–60. doi: 10.1001/jamafacial.2015.1558.
- Biglioli F, Colombo V, Rabbiosi D, Tarabbia F, Giovanditto F, Lozza A, Cupello S, Mortini P. Masseteric-facial nerve neurorrhaphy: results of a case series. J Neurosurg. 2017;126(1):312–318. doi: 10.3171/2015.12.jns14601.
- Hontanilla B, Marré D. Comparison of hemihypoglossal nerve versus masseteric nerve transpositions in the rehabilitation of short-term facial paralysis using the Facial Clima evaluating system. Plast Reconstr Surg. 2012;130(5):662–672. doi: 10.1097/PRS.0b013e318267d5e8.
- Zhang S, Hembd A, Ching CW, Tolley P, Rozen SM. Early masseter to facial nerve transfer may improve smile excursion in facial paralysis. Plast Reconstr Surg Glob Open. 2018;15;6(11):e2023. doi: 10.1097/GOX.0000000000002023.
- Vila PM, Kallogjeri D, Yaeger LH, Chi JJ. Powering the gracilis for facial reanimation: A systematic review and meta-analysis of outcomes based on donor nerve. JAMA Otolaryngol Head Neck Surg. 2020;146(5):429–436. doi: 10.1001/jamaoto.2020.0065.
- Boahene KO, Owusu J, Ishii L, Ishii M, Desai S, Kim I, Kim L, Byrne P. The multivecto gracilis free functional muscle flap for facial reanimation. JAMA Facial Plast Surgery. 2018;20(4);300–306. doi: 10.1001/jamafacial.2018.0048.
- Viterbo F. Secondary procedures in facial reanimation. In: Grotting JС, editor. Reoperative aesthetic & reconstructive plastic surgery. 2nd ed. St. Louis, Missouri: Qualty Medical Publishing; 2007. р. 859–879.
- Kim MJ, Kim HB, Jeong WS, Choi JW, Kim YK, Oh TS. Comparative study of 2 different innervation techniques in facial reanimation cross-face nerve graft-innervated versus double-innervated free gracilis muscle transfer. Ann Plast Surg. 2020;84(2):188–195. doi: 10.1097/sap.0000000000002034.
- Park Seong Oh, Ha Jeong Hyun, Kim Il‐Kug, Jin Ung Sik, Chang Hak. Single‐stage cross‐facial nerve grafting has a result equivalent to that of two‐stage cross‐facial nerve grafting. Microsurgery. 2020;40(2):175–182. doi: 10.1002/micr.30448.
Supplementary files